DENV Detect™ IgM Capture ELISA Kit
FDA Cleared for In Vitro Diagnostic Use
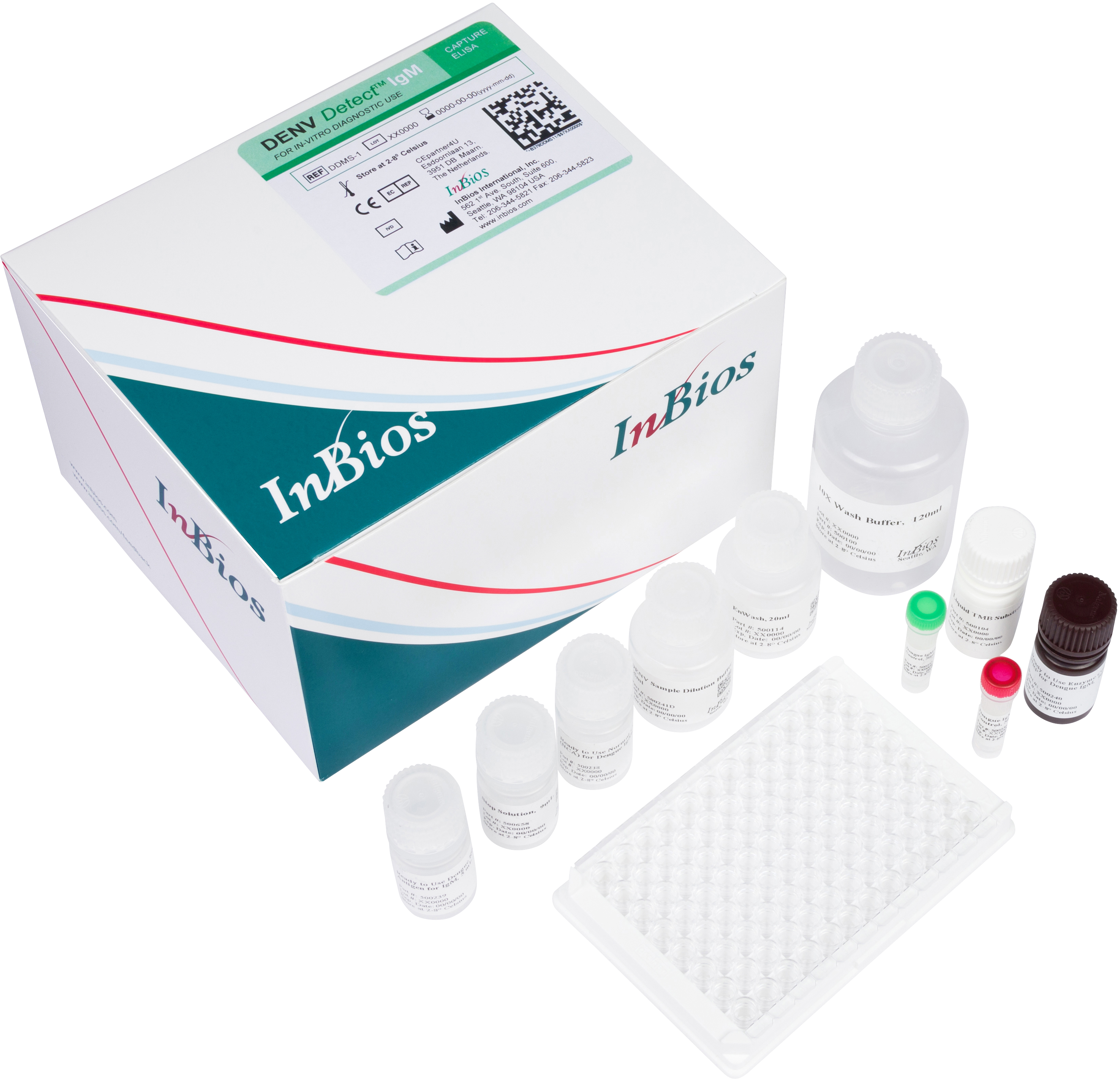
The DENV Detect™ IgM Capture ELISA is for the qualitative detection of IgM antibodies to DENV recombinant antigens in serum for the presumptive clinical laboratory diagnosis of Dengue virus infection.

Resources
1st Assay to Receive FDA Clearance
Performance thoroughly evaluated with clinically confirmed cases of dengue 1-4 serotypes
Excellent positive and negative agreement with WHO reference panel (>90%)
Improves accuracy by monitoring background reactivity with a normal cell antigen
Employs a simple, one step ratio method of interpretation
CE Marked
| Catalog No. | DDMS-1 |
| Format | Immunocapture |
| Quantity/Kit | 96 wells/plate |
| Incubation Time | 60+60+60+5+10+1 |
| Shelf Life | 12 months |
| Sample Type | Serum |
| Storage | 2-8°C |
For more information, download instructions for use via the “Downloads” tab.
Related Products
ZIKV Detect™ 2.0 IgM Capture ELISAinfo@inbios.com2023-10-11T21:39:09+00:00
ZIKV Detect™ 2.0 IgM Capture ELISA
Scrub Typhus Detect™ IgG ELISAinfo@inbios.com2023-10-31T15:14:44+00:00
Scrub Typhus Detect™ IgG ELISA
JE Detect™ IgM Antibody Capture ELISA for Japanese Encephalitisinfo@inbios.com2023-10-11T21:43:13+00:00
JE Detect™ IgM Antibody Capture ELISA for Japanese Encephalitis
Scrub Typhus Detect™ IgM ELISAinfo@inbios.com2023-10-11T21:44:00+00:00