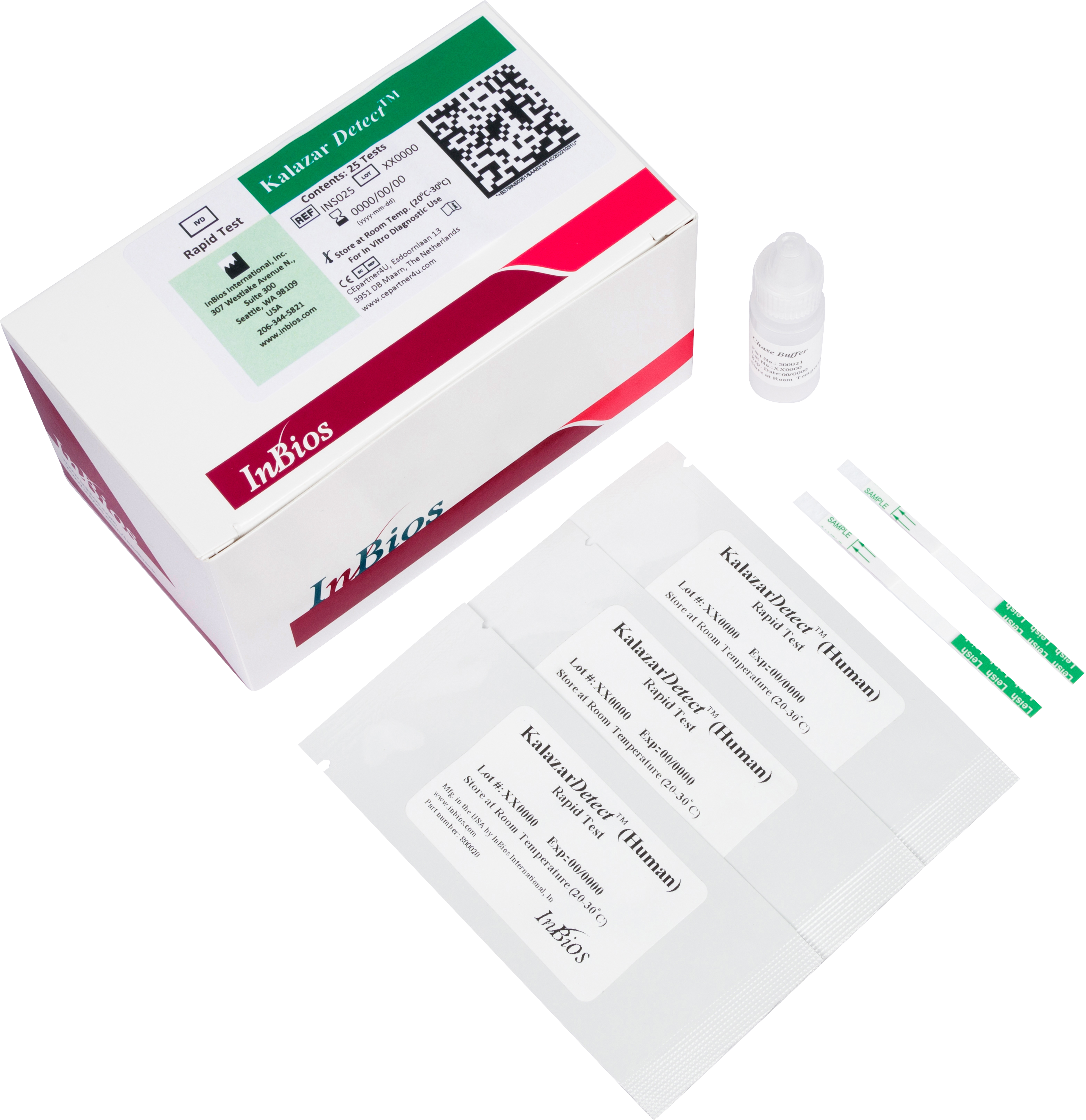
 Kalazar Detect™ Rapid Test for Visceral Leishmaniasis
Kalazar Detect™ Rapid Test for Visceral Leishmaniasis
FDA Cleared for In Vitro Diagnostic Use
The Kalazar Detect™ Rapid Test for Visceral Leishmaniasis (VL) is a rapid immunochromatographic strip assay for the qualitative detection of antibodies to members of L. donovani in human serum to aid in the presumptive diagnosis of VL. This kit is intended for professional in vitro diagnostic use only. It is not intended for use in blood donor centers or by blood component manufacturers.
- US FDA Cleared
- CE Marked
- Sensitivity and Specificity: >90%
- Fast Results: 10 minutes
- Field Friendly: Refrigeration and lab equipment not required
| Catalog No. | Format | Quantity/Kit | Time to Result | Sample Type | Storage | Shelf Life |
|---|---|---|---|---|---|---|
| INS025 | ICT (strip) | 25 Tests | 10 minutes | Serum | Room Temperature (20℃ - 30℃) | 24 months |
